One-stop POCT Medical Diagnostic Platform
The I Holdings is the world’s only medical diagnostic platform provider to introduce the new innovative virus-controlled modular structure (The I Dove™), cutting-edge diagnostic testing kits, medical products, and a systematic universal operation solution (IDX solution) all together to provide the optimal POC environment.
To develop the best Point-of-Care environment that fulfills all of our client’s requests, we provide the following services that consist of professional consultation, R&D procedure, module customization, and launch & operation planning. Through these services, we build the ultimate go-to One-stop On-site Medical Diagnostic Testing Center that can perform all stages from specimen collection to isolation of confirmed cases. The best part is that we can execute all procedures without the constraint of time and space.
Our unique platform business is a fast, accurate, convenient, and safe medical diagnostic solution that will boost the current medical infrastructure and save more lives during all types of medical and diagnostic emergencies.
The One-stop POCT Center
Integrated Service for Medical
Diagnostic Products
Customization Service
for Optimized POC environment
One-stop On-site
Medical Diagnostic Platform
Core Strategies
Win-Win Proposition
-Procurement of stable supply chain system
Global Reach
-Researchers with high proficiency in R&D for disease diagnosis solutions
-Ability to provide appropriate on-site medical diagnostic solutions
Expertise
-Strategic development of environmentally optimized products to satisfy market demands
-Superior global networking skills for successful overseas expansion
-Attainment of competent, unique global partnerships
-Efficient coordination with accredited organizations around the world
* Ownership of FDA · EUA channel
* Partnership with FDA approved CLIA Labs
IDX Center, On-site Diagnostic Testing Center
Recent events dealing with COVID-19 and its variants have led to skyrocketing demand for fast on-site (POCT) diagnosis and medical treatment. We created the IDX Center, a total diagnostic service center, to conduct prompt, accurate, and safe on-site diagnosis to efficiently act upon all market’s needs. All users of the IDX Center will receive the full package that includes The I Dove™, a virus-controlled on-site medical diagnostic module, with the IDX Solution, operation manual of the IDX Center.
IDX Solution
+
The I Dove™
=
IDX Center
IDX Solution
IDX Solution provides the best operation solution for POC medical diagnostic center that is suitable for pandemic situations and satisfies the needs of global consumers. It is not only based off of our deep knowledge and accumulated experience dealing with regulations, but also complies with WHO’s operational guidelines and country-specific regulations.
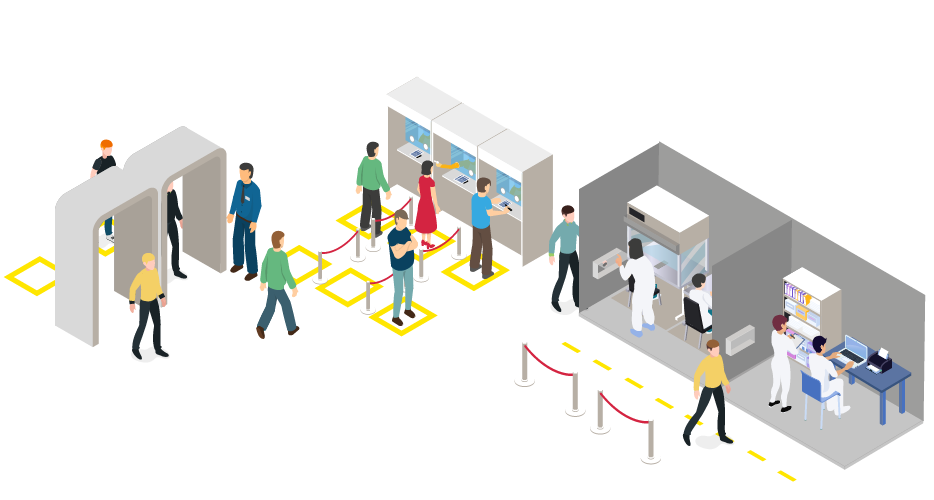
| COVID19 Operation Flow of COVID-19 Medical Diagnostic Center | Questionnaire, Fever Check | → | Specimen Collection | → | Specimen Pre-treatment
(RNA extraction or buffer mix) |
→ | Diagnostic test (Molecular or Antigen) | → | Issuing of Test Result |
| Operation Flow of COVID-19 Medical Diagnostic Center |
| 1. Questionnaire, Fever Check |
| 2. Specimen Collection |
| 3. Specimen Pre-treatment (RNA extraction or buffer mix) |
| 4. Diagnostic test (Molecular or Antigen) |
| 5.Issuing of Test Result |
The I Dove™_On-site Medical Diagnostic Modules
The I Dove™ is a virus-controlled on-site medical diagnostic module optimized for the POC environment. It can be customized based on client’s needs and preferences in terms of shape, size, and structural design. Its strategic design guarantees speedy construction and set up at the designated location that urgently needs on-site diagnosis or medical treatment.
On-site Diagnostic Modules

The I Dove™_Module Type

The I Dove™_ Pre-fabricated Type

The I Dove™ _Movable Lab Type
On-site Medical Modules

The I Dove™_SNPIR
(Simple Negative Pressure Isolation Room)

The I Dove™_MIS
(Medical Isolation Studio)

The I Dove™_ICU
(Intensive Care Unit)
Professional Integrated Service for Medical Diagnostic Products
Counseling for Diagnostic Products
1. Support to obtain country-specific
certifications and permissions
1. Support to obtain country-specific certifications and permissions
-FDA-EUA for USA, CE for Europe, DHSC for UK
-FDA sales permission for Saudi Arabia, and more.
-Assist with document submission and follow up with certification process
2. Help set up launching strategy by country
– Upon completion of technical review, select the appropriate country to target and advise on launch strategies
2.
Technological review of COVID-19
diagnostic products
1. Diagnostic testing kit: RT-qPCR, RT-LAMP, dPCR, Antigen, Antibody, Biosensor
2. Specimen collection tools: Swab, Mask Patch, Saliva Collection
3. Nucleic acid extraction solution: Solution for RNA extraction without using a machine
4. LAMP machine: Optimal device for RT-LAMP test (16 wells)
Custom-made R&D support
Customizing diagnostic products according to
customer's needs
1. Customization of The I Dove™’s size, shape, and type before delivery and installation
2. Supply chain of various types of COVID-19 diagnostic kit, machines and more
3. Many simple medical equipment available for POC environment
New Product Development based on
Current Market Trends
1. Ability to rapidly develop medical diagnostic products during pandemic times
2. Continuous analysis of global market trends such as Point of Care Testing (POCT), non-face-toface telemedicine, IOT, Wearable, and more
Customization Service for POCT environment
We provide customized modules, diagnostic kits/ equipment, medical devices, and operational plans to configure a POCT environment suitable for the consumer’s circumstances and purpose of operation
Custom-made POCT
➊ POCT for Incheon Int’l Airport T1 for airport users
(partnered with Iwon Medical Foundation, Installed August 2021~Present)
➋ POCT for Bestian Osong Hospital
(partnered with KGIC. Ready to open in Jan. 2022)
➌ POCT for international airport in Thailand
➍ POCT for Malaysian border area
➎ Establishment and operation of an on-site quarantine system during the Qatar World Cup
(Airport, government office, soccer stadium)
➏ POCT for general hospitals in Saudi Arabia
Recommending the Right Product
(Modules, diagnostic kits/equipment, medical equipment, and other)
➊ Link with CLIA Lab in the US
– Contract to supply LDT Antigen Kit for POC purpose
– Clinical trials for one type of RT-LAMP and one type of RT-qPCR to go under review
➋ Prepare operation & establishment plan of US IDX Center with US partner
➌ Propose to UK partner diagnostsic testing kits for POC and/or research use
➍ Prepare POCT center in Europe (e.g. Germany)
Customization Service for POCT environment
We provide customized modules, diagnostic kits/ equipment, medical devices, and operational plans to configure a POCT environment suitable for the consumer’s circumstances and purpose of operation
Custom-made POCT
➊ POCT for Incheon Int’l Airport T1 for airport users
(partnered with Iwon Medical Foundation, Installed August 2021~Present)
➋ POCT for Bestian Osong Hospital
(partnered with KGIC. Ready to open in Jan. 2022)
➌ POCT for international airport in Thailand
➍ POCT for Malaysian border area
➎ Establishment and operation of an on-site quarantine system during the Qatar World Cup
(Airport, government office, soccer stadium)
➏ POCT for general hospitals in Saudi Arabia
Recommending the Right Product
(Modules, diagnostic kits/equipment, medical equipment, and other)
➊ Link with CLIA Lab in the US
– Contract to supply LDT Antigen Kit for POC purpose
– Clinical trials for one type of RT-LAMP and one type of RT-qPCR to go under review
➋ Prepare operation & establishment plan of US IDX Center with US partner
➌ Propose to UK partner diagnostsic testing kits for POC and/or research use
➍ Prepare POCT center in Europe (e.g. Germany)
